Late winter in New England is prime time for respiratory bugs to circulate — especially influenza (flu), RSV, COVID-19, and “common cold” viruses. National monitoring in February 2026 showed flu activity remained elevated, with influenza B increasing, and RSV elevated in some areas. In a recent epidemiology report, RSV was at peak winter levels statewide, and influenza activity remained elevated, with the highest burden in young children and older adults.
Because many respiratory illnesses share the same symptoms, it can be hard to tell what you have based on how you feel alone. Testing and good “stay-home” habits help protect your family, coworkers, classmates, and community.
What’s Going Around in Winter 2026?
In most winters, the biggest drivers of respiratory illness are:
- Flu
- RSV (Respiratory Syncytial Virus)
- COVID-19
- Common cold viruses (like rhinovirus)
These viruses spread mainly through close contact, respiratory droplets/aerosols, and hands/surfaces — which is why indoor season and gatherings can fuel outbreaks.
Common Symptoms (and why they overlap)
Respiratory viruses can cause a mix of upper-airway and whole-body symptoms, including:
- Runny/stuffy nose, sore throat
- Cough
- Fever or chills (not always)
- Body aches, headache, fatigue
Flu often comes on suddenly and can cause significant body aches and fatigue.
RSV often looks like a cold at first, but it can be more serious for infants, older adults, and people with certain health conditions.
COVID-19 symptoms range from mild to severe, and can overlap with flu/RSV.
People Who Should Take Extra Precautions
Some groups are more likely to get very sick from respiratory viruses, including older adults, young children, pregnant people, and people with certain medical conditions or weakened immune systems.
When to Stay Home
CDC’s current respiratory virus guidance recommends:
Stay home and away from others until:
- Your symptoms are improving overall, and
- If you had a fever, you’ve been fever-free for at least 24 hours without fever-reducing medicine.
After you return to normal activities:
For the next 5 days, take extra precautions (like masking and improving ventilation), because you may still be able to spread the virus even as you’re feeling better.
If you start to feel worse again or your fever returns:
Go back to staying home and restart the “improving + 24 hours fever-free” clock.
When to Test
Testing can help you:
- Make safer choices about work/school/visiting others
- Protect high-risk family and friends
- Decide whether you might qualify for early treatment
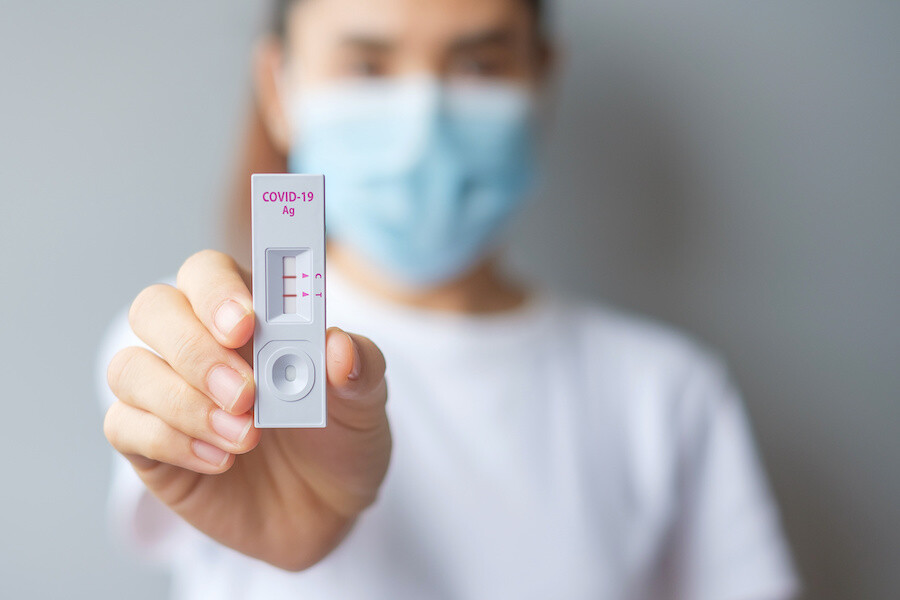
Test Right Away if You Have Symptoms
COVID-19: If you use an at-home antigen test and it’s negative, that doesn’t always rule out infection—especially early on. CDC advises following the FDA guidance for repeat testing after a negative antigen result.
Flu and COVID: If you’re at higher risk for complications, earlier diagnosis matters because treatments work best when started quickly. (More on that below.)
When a Negative Test Shouldn’t Reassure You
Consider retesting or calling a clinician if:
- Symptoms are getting worse
- You tested very early after symptoms began
- You have close exposure and symptoms
- You’re high-risk or live with someone high-risk
When to Seek Care
Call your healthcare provider sooner (same day if possible) if:
- You’re in a higher-risk group (older adult, pregnancy, chronic conditions, immunocompromised, etc.)
- Symptoms are worsening after initial improvement
- Fever is persistent, or you’re worried about dehydration
- You may be eligible for antiviral treatment (flu/COVID)
Seek Emergency Care Right Away if You Notice Warning Signs Like:
For COVID-19: trouble breathing, persistent chest pain/pressure, new confusion, inability to wake/stay awake, or pale/gray/blue lips/nail beds (depending on skin tone).
For flu: CDC lists emergency warning signs for adults and children (including breathing difficulty, chest/abdomen pain or pressure, confusion, seizures, dehydration/not urinating, or symptoms that improve then return/worsen).
For RSV: call a healthcare professional if you or your child has difficulty breathing, isn’t drinking enough fluids, or symptoms are worsening.
What to Do if You Think You Have a Respiratory Illness
For most mild illnesses:
- Rest, hydrate, and use over-the-counter fever/pain reducers as appropriate
- Stay home until you meet the “improving + 24 hours fever-free” rule
- Avoid close contact with people at higher risk
Ask About Treatment Early if You’re at Higher Risk
- Flu antivirals can lessen symptoms and shorten illness; they work best when started within 1–2 days of symptom onset and are especially important for people at higher risk of complications.
- Outpatient COVID-19 treatment for eligible patients should be initiated as soon as possible, ideally within a few days of symptom onset (depending on the medication).
How to Help Prevent the Spread
CDC’s prevention approach emphasizes layered protection:
Stay up to date on recommended immunizations (flu, COVID-19, and RSV for eligible groups).
- RSV vaccine guidance (adults) and maternal RSV vaccination guidance (pregnancy) are available through the CDC.
Improve indoor air when possible: bring in outdoor air, use filtration, or gather outdoors.
Wash hands, cover coughs/sneezes, and clean high-touch surfaces.
If you must be around others while recovering, mask and keep distance—especially around older adults, infants, and medically fragile people.